
An ideal gas is a theoretical gas composed of many randomly moving point particles that do not interact except when they collide elastically. The ideal gas law is the equation of state of an ideal gas. It relates the state variables of the gas: pressure

A New Physics Theory of Life
upload.wikimedia.org/wikipedia/commons/thumb/c/cd/

Boltzmann distribution - Wikipedia

Mechanism of sonoluminescence - Wikipedia

Phase diagram - Wikipedia

Kinetic Theory of an Ideal Gas: Equation, Assumption, Concept, Examples

Boyle's law - Wikipedia

Law of total expectation - Wikipedia

Ideal Gas Law, (PV=nRT), P is the pressure of the gas, V is the volume of the gas, n is the amount of s…
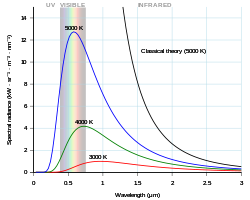
Planck's law - Wikipedia
Is a bonding molecular orbital more stable than an antibonding molecular orbital? Why? - Quora






