Microbiological Media Management - SOP & Guideline - Pharma Beginners

Standard Operating Procedure (SOP) and Guideline for the Receipt, Storage, Preparation, Growth Promotion Test, use, and Disposal of microbiological media.

SOP for Isolation and Identification of Microorganisms - Pharma Beginners
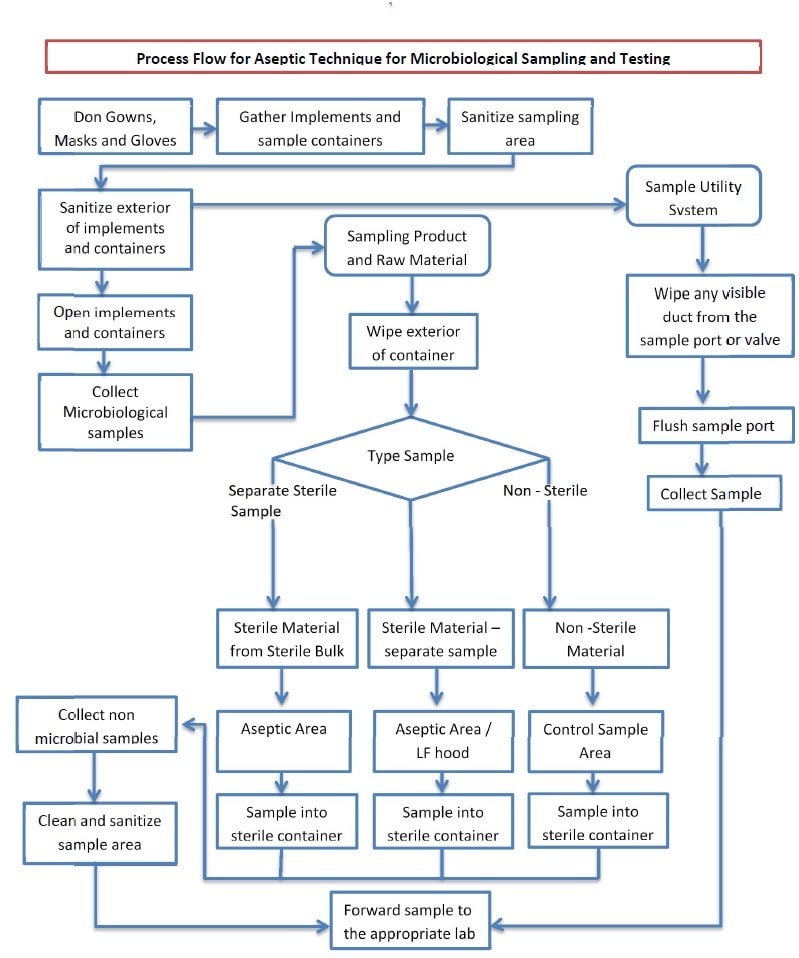
Aseptic Technique for Microbiological Testing - Pharma Beginners

Validation of Aseptic Processes Using Media Fill
The importance of growth promotion testing

Stability testing for pharmaceutical products by GMP SOP - Issuu

Sterility Test, how to perform Sterility Test in the best way?

Microbiology Quality Control Testing: Definition & Procedures

PDF) Microbiological Culture Media: A Complete Guide for Pharmaceutical and Healthcare Manufacturers
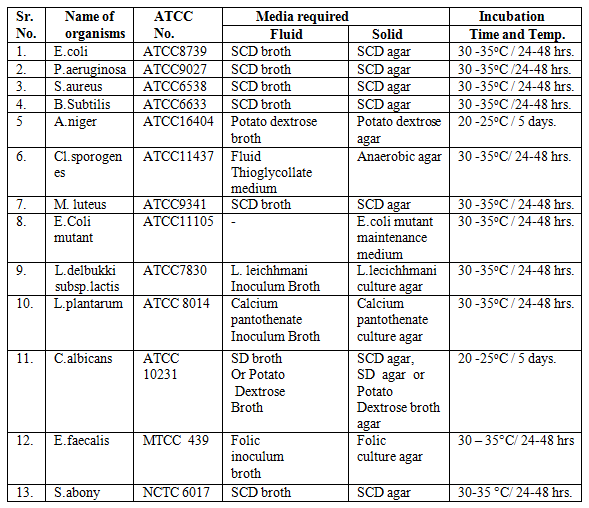
SOP for Maintenance and Transfer of Stock Cultures : Pharmaguideline

PDF) Preparing SOP for Microbiology Laboratory: A Short Guideline
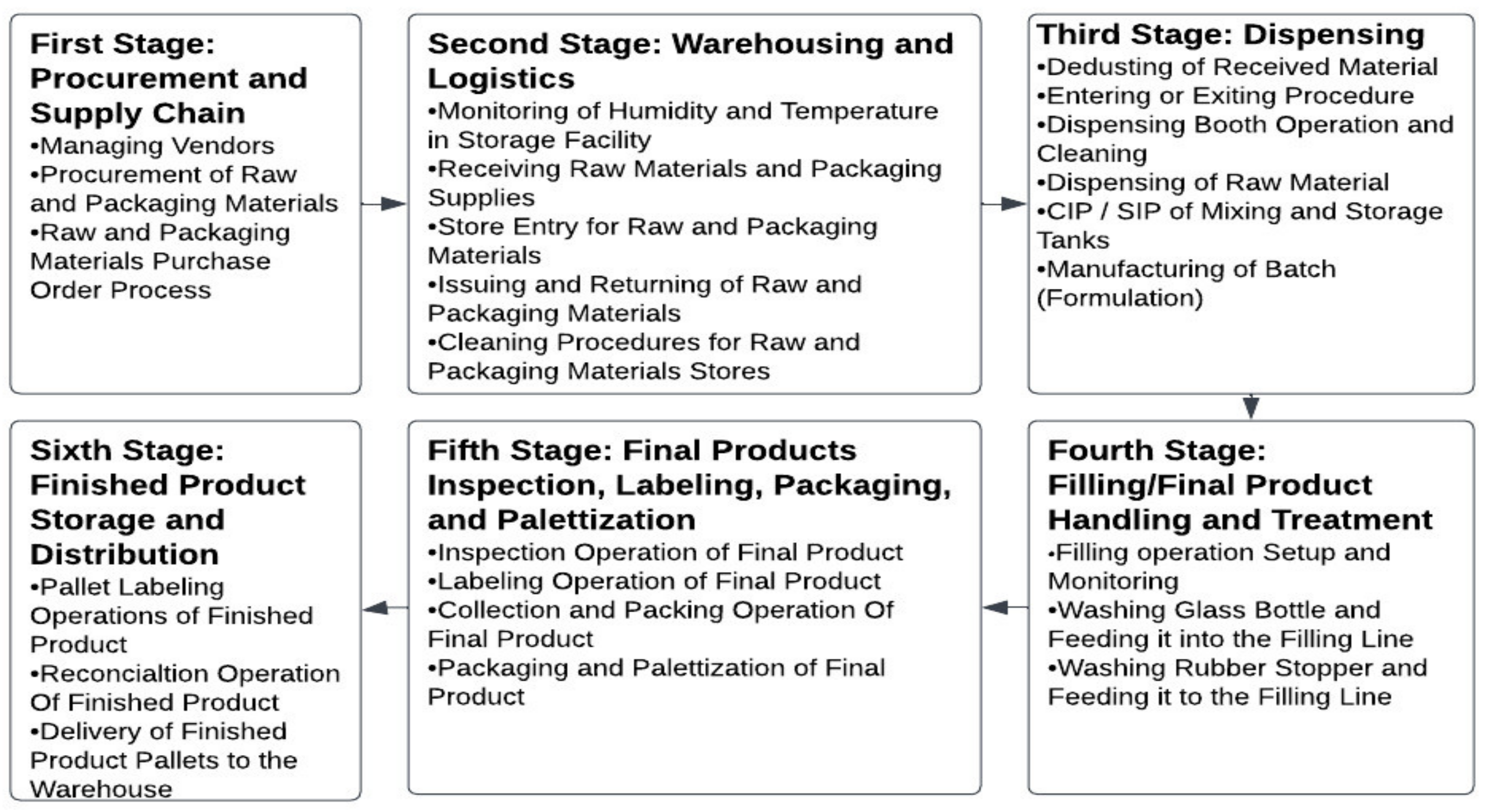
Sustainability, Free Full-Text

Resume Advice Appreciated : r/biotech







