Structure of YF1. a Structure of YF1 in its dark-adapted state as

Download scientific diagram | Structure of YF1. a Structure of YF1 in its dark-adapted state as resolved by X-ray crystallography 13. The location of the different domains, of the flavin mononucleotide (FMN), of the cofactor adenosine diphosphate (ADP), and of the phosphoaccepting histidine 161 are indicated. b Light induced conformational changes of the LOV photosensor domain refined from X-ray solution scattering 22. The changes are maximal at the C-termini that feed into the Jα helices (dashed arrows). The coloring is according to the root mean square deviation of the alpha carbons from publication: Sequential conformational transitions and α-helical supercoiling regulate a sensor histidine kinase | Sensor histidine kinases are central to sensing in bacteria and in plants. They usually contain sensor, linker, and kinase modules and the structure of many of these components is known. However, it is unclear how the kinase module is structurally regulated. Here, we use | Secondary Protein Structure, Bacterial Proteins and Protein Conformation | ResearchGate, the professional network for scientists.

Ontogeny of the Postorbital Region in Tarsiers and Other Primates - DeLeon - 2016 - The Anatomical Record - Wiley Online Library

Crystal structure of LOV-PAS and LOV-PAS-HK. (A) Ribbon diagram of

Structure of the LOV domain of YF1 (PDB entry 4GCZ (17)). Secondary

Signal transduction in light–oxygen–voltage receptors lacking the adduct-forming cysteine residue

Rapid evolutionary repair by secondary perturbation of a primary disrupted transcriptional network
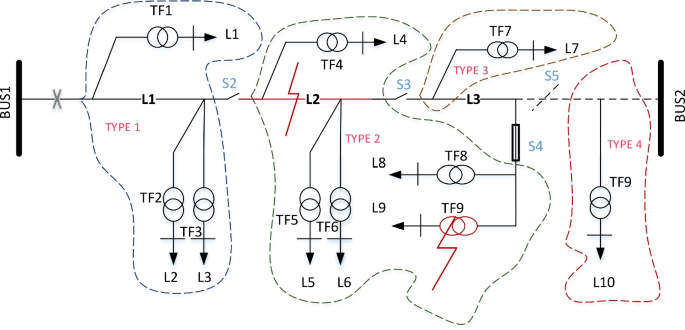
Power distribution system reliability evaluation using weight-optimised ANN approach

Maria HOERNKE, Postdoctoral Fellow, PhD in physical chemistry, University of Freiburg, Freiburg, Albert-Ludwigs-Universität Freiburg, BIOSS Centre for Biological Signalling Studies

Matthijs PANMAN, Scientist, PhD, VSL - Dutch Metrology Institute, Delft, VSL, Temperature and Humidity

S-band FSE spectrum of YtvA T54C/C176S/T179C (solid line) and YtvA