The Smallest Artificial Pancreas System Receives FDA Clearance - JDRF
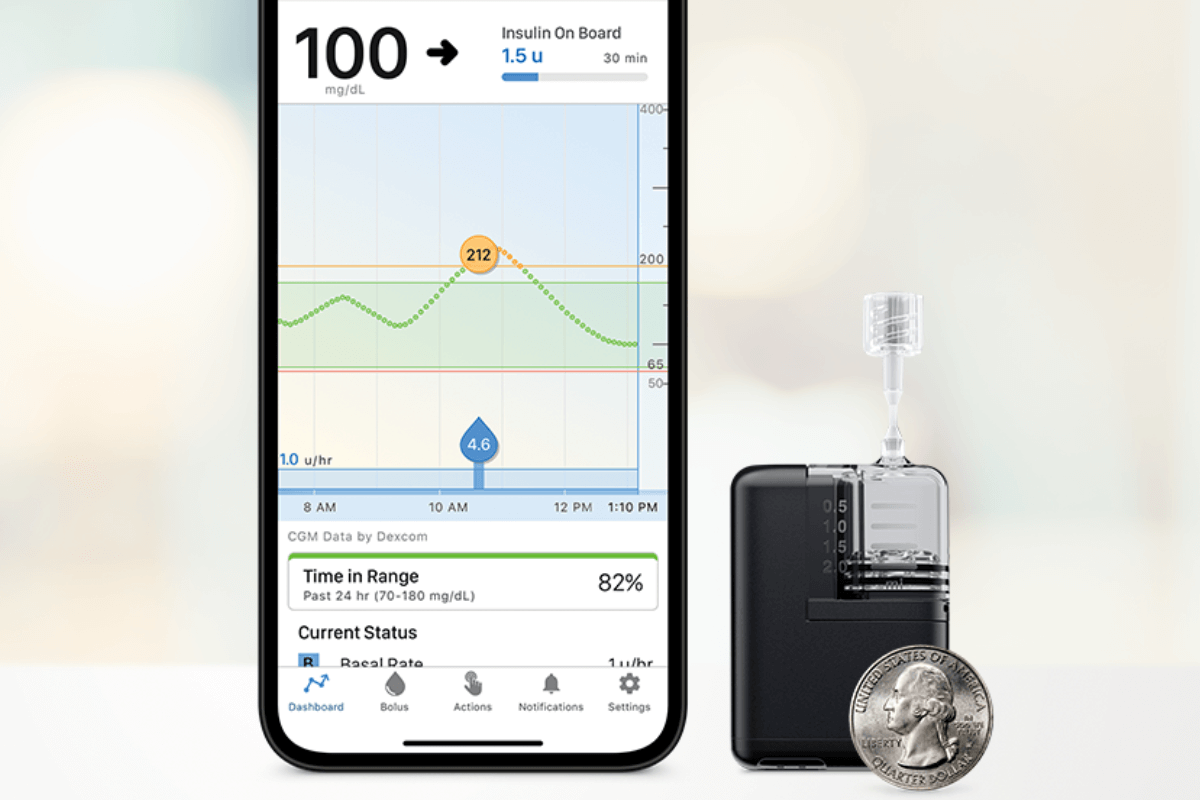
In just the past year, we have had multiple artificial pancreas systems authorized by the Food and Drug Administration (FDA)…and it’s not stopping! Last week, Tandem Mobi—a miniature-sized insulin pump, for use with Tandem’s Control-IQ™ technology and a compatible continuous glucose monitor (CGM)—received FDA clearance. The Tandem Mobi is half the size of the company’s…

Models, Devices, Properties, and Verification of Artificial
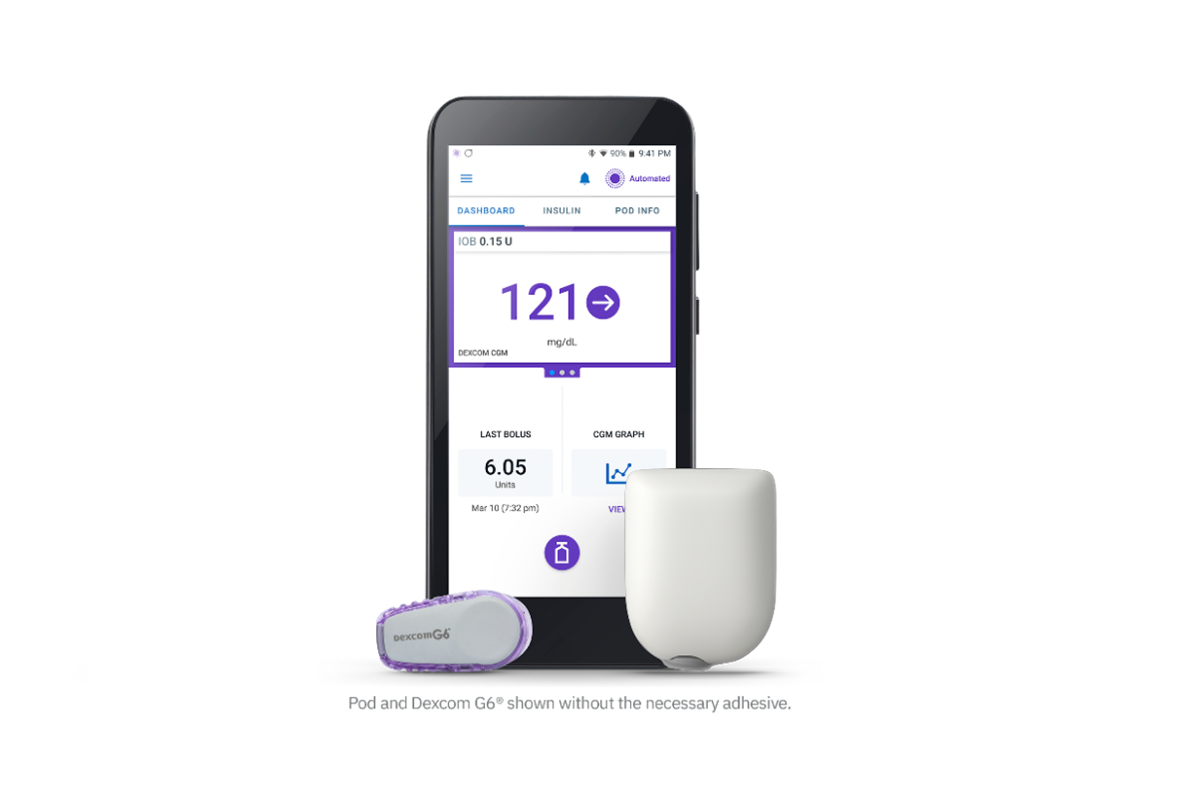
FDA Authorizes a Fourth Artificial Pancreas System: JDRF Celebrates

i0.wp.com/post./wp-content/uploads/2

Looping' Created an Underground Insulin-Pump Market - The Atlantic
Courtney Lias, Stayce Beck and the FDA Artificial Pancreas Team • Samuel J. Heyman Service to America Medals

Hoping to make big tracks, Bigfoot Biomedical scores FDA clearance

World's First 'Artificial Pancreas' Approved for Treatment of Type

Hoping to make big tracks, Bigfoot Biomedical scores FDA clearance
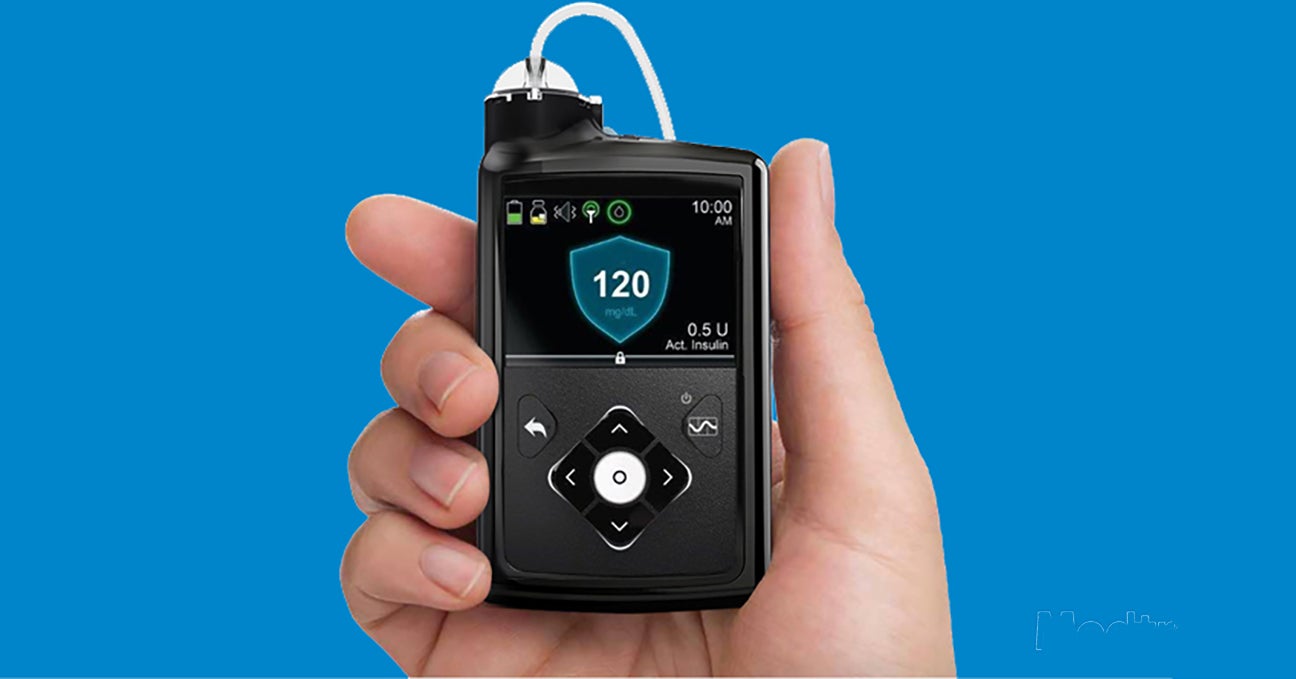
Artificial Pancreas aka Automated Insulin Delivery: What You Should

Glucose-responsive insulin delivery for type 1 diabetes: The

Bigfoot Biomedical® Receives FDA Clearance for Bigfoot Unity™ Diabetes Management System, Featuring First-of-its-Kind Smart Pen Caps for Insulin Pens used to Treat Type 1 and Type 2 Diabetes