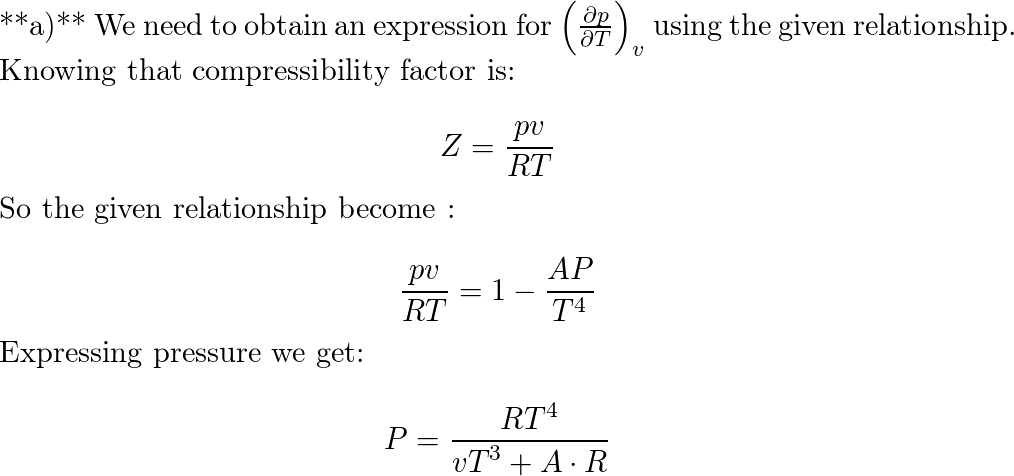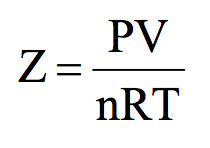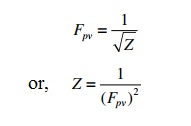20.If Z is a compressibility factor, van der Waals equation at low pressure can be written as
20.If Z is a compressibility factor, van der Waals equation at low pressure can be written as
20-If Z is a compressibility factor- van der Waals equation at low pressure can be written as

Solved Can you solve the problem and add fortran code for

Compressibility Factor Calculator - File Exchange - MATLAB Central

At low pressure, the compressibility factor is given as

Solved Problem 2. ( 30 points) The ideal gas law can

Solved 2. (20 points) At low pressures, the compressibility

Deviations from ideal gas behaviour, intermolecular forces, Van der Waals equation of state, compressibility factors and the critical pressure and critical temperature of a gas revision notes doc brown's chemistry UK advanced

1. The ideal gas law can represent the
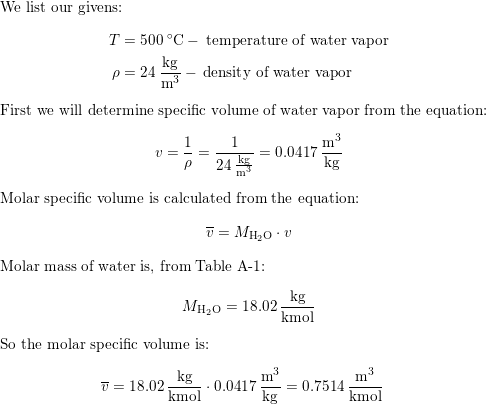
Estimate the pressure of water vapor at a temperature of $50

If Z is a compressibility factor, van der Waals equation at low pressure can be written as [JEE

Solved) - For values of z near 1, it is a good approximation to write z(P) = - (1 Answer)

Compressibility factor (Z) for a van der Waals real gas at critical po

Solved The Van Der Waals equation of state is given by
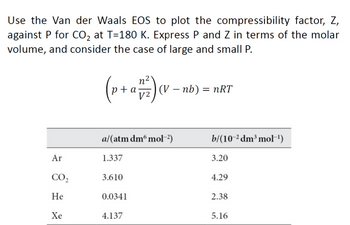
Answered: Use the Van der Waals EOS to plot the…

1.7: Connecting the van der Waals and the viral equations- the Boyle temperature - Chemistry LibreTexts

Solved) - For values of z near 1, it is a good approximation to write z(P) = - (1 Answer)