Click here:point_up_2:to get an answer to your question :writing_hand:an ideal gas initially at pi vi and ti is taken through a cycle
Click here👆to get an answer to your question ✍️ An ideal gas initially P-i -V-i - and T-i is taken through a cycle as shown in Figure- -a- Find the net work done on the gas per cycle 1-00 mol of gas initially 0-0C- -b- What is the net energy added by heat to the gas per cycle

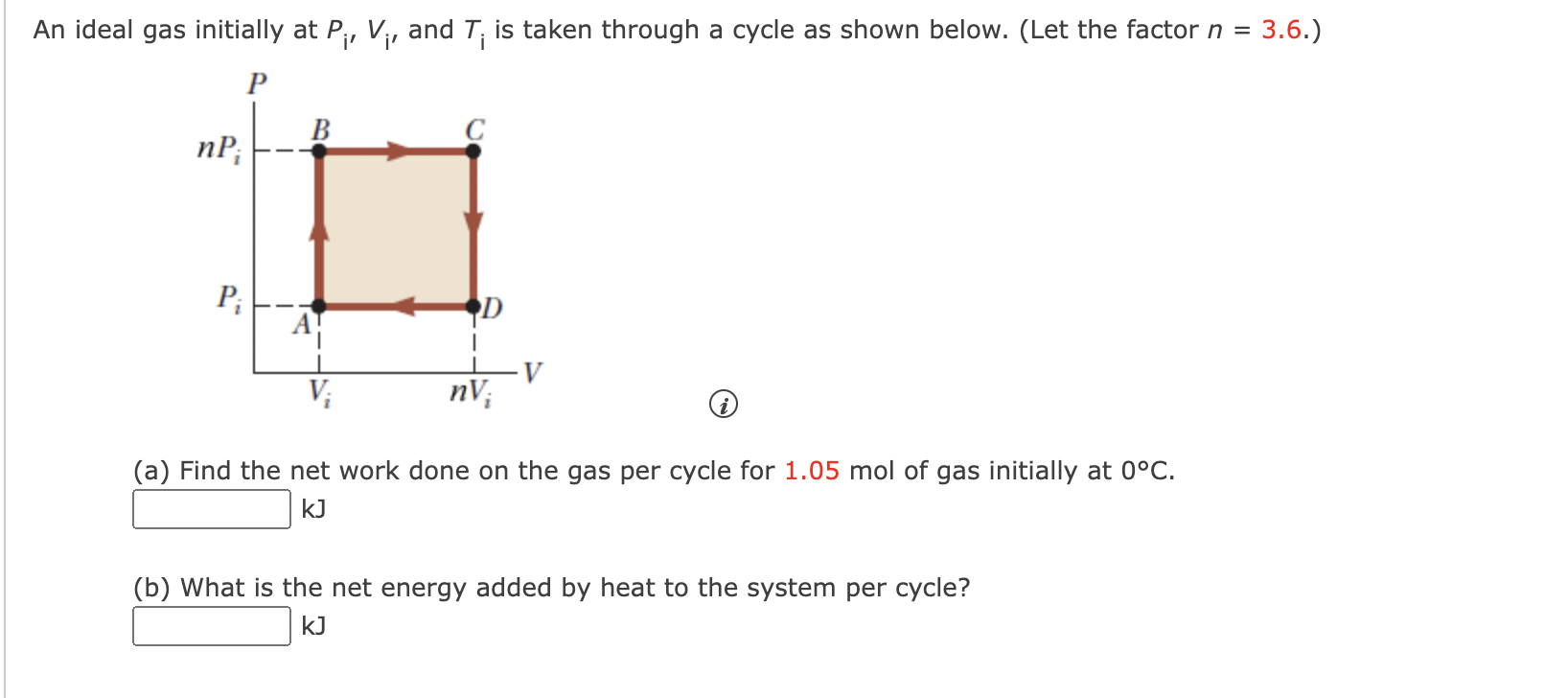
SOLVED: An ideal gas initially at Pi, Vi, and Ti is taken through a cycle as shown below. (Let the factor n 3.6.) nP; P; nV; (a Find the net work done
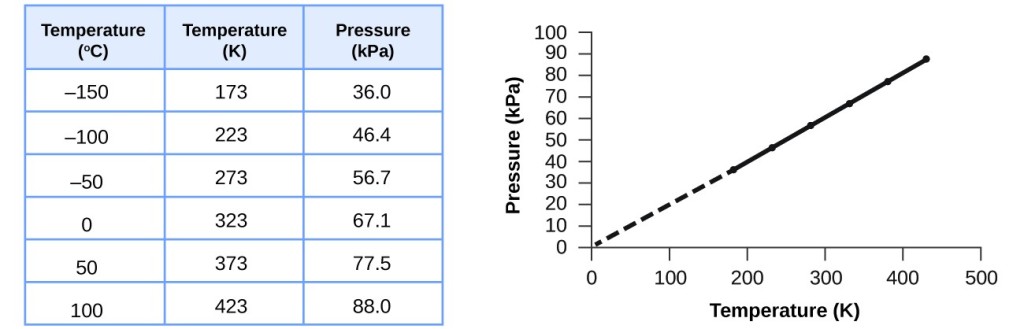
Relating Pressure, Volume, Amount, and Temperature: The Ideal Gas Law

One mole of a gas in the state A(P1, V1, T1) is subjected to adiabatic

Chapter4, Lesson F - Performance of an Ideal Gas Cycle
Chemosensors, Free Full-Text

SOLVED: An ideal gas initially at Pi' Vi' and Ti is taken through a cycle as shown below. (Let the factor n 2.8.) nP; nV; a) Find the net work done on

P) Thermodynamics, PDF, Gases

Thermodynamics

Entropy of a Gas
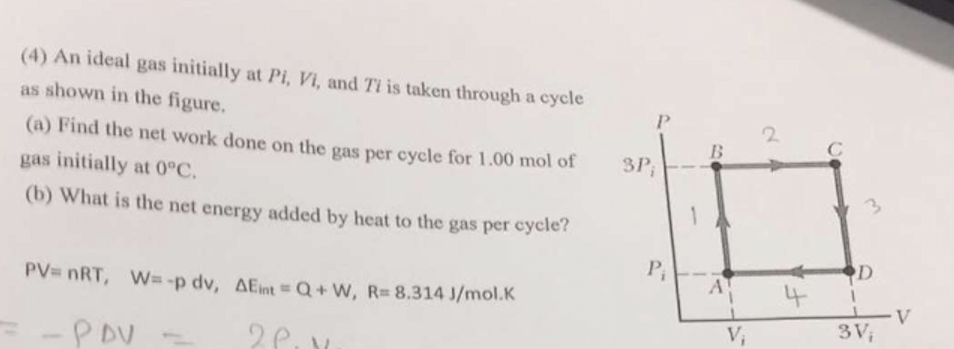
Solved (4) An ideal gas initially at Pi, Vi, and Ti is taken
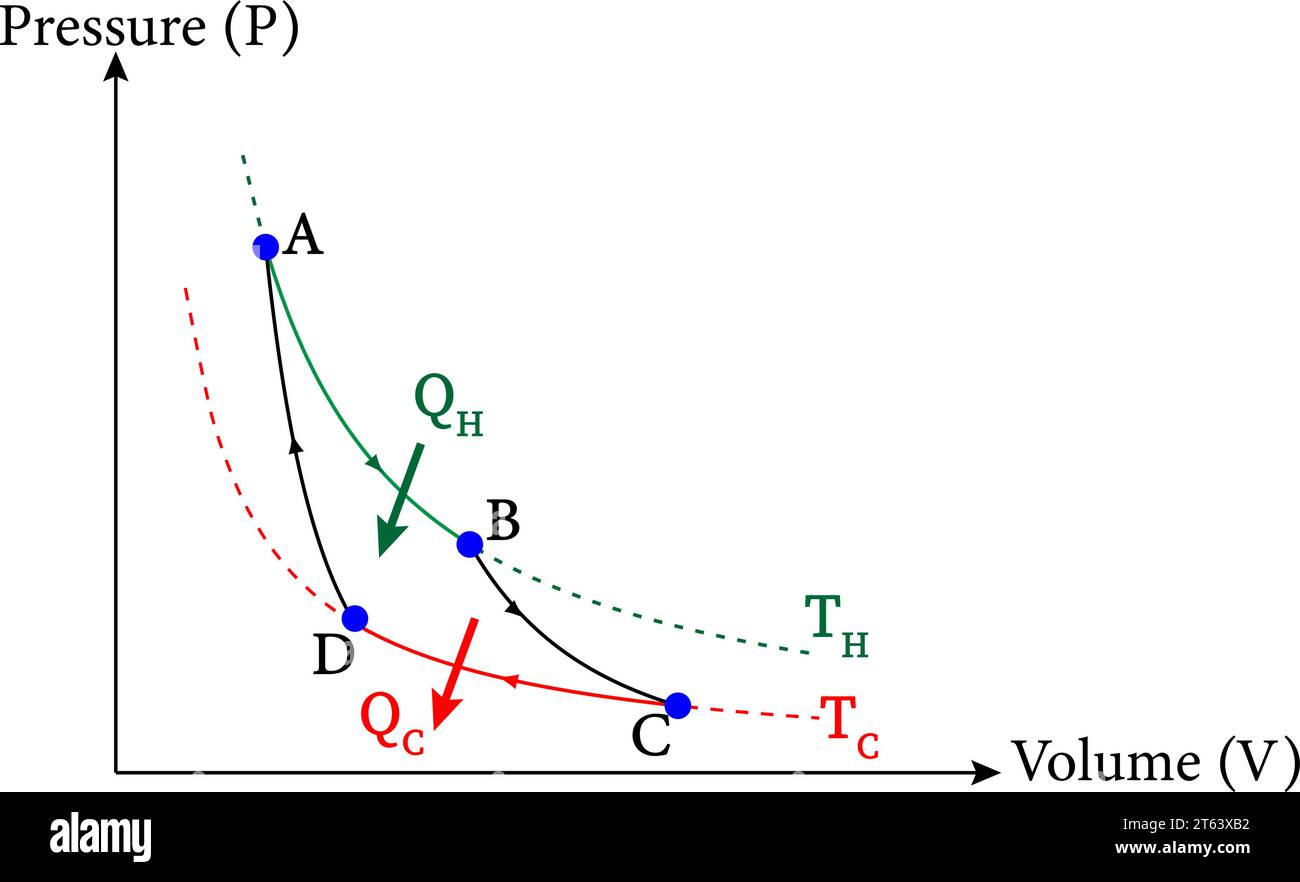
Carnot cycle hi-res stock photography and images - Alamy
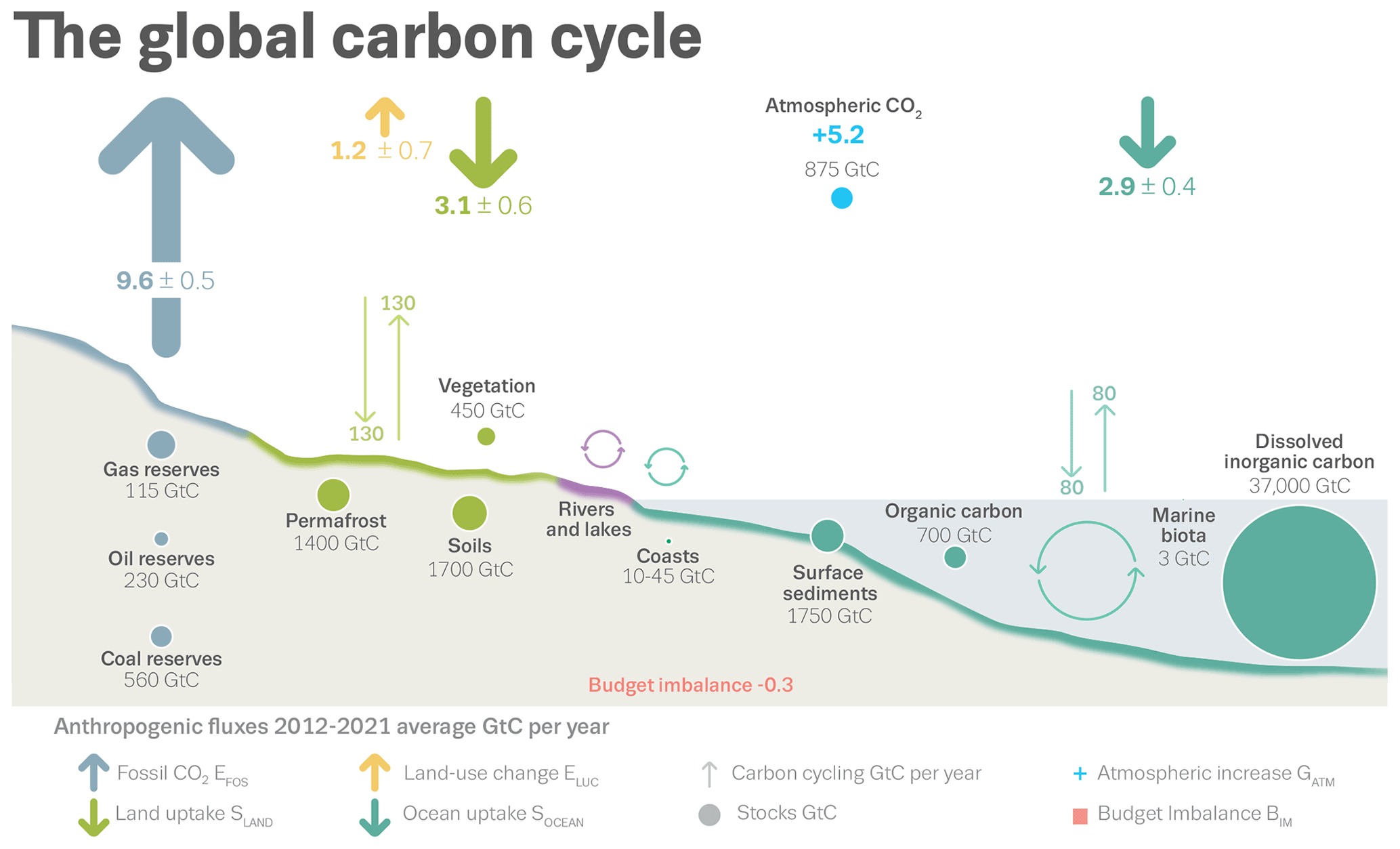
ESSD - Global Carbon Budget 2022

An ideal gas initially at Pi, Vi, and Ti, is taken through a cycle as shown below. (Let the factor n= 3.6.) a. Find the work done on the gas per cycle

Process integration, energy and exergy analyses of a novel integrated system for cogeneration of liquid ammonia and power using liquefied natural gas regasification, CO2 capture unit and solar dish collectors - ScienceDirect

An ideal gas is taken through the cycle A → B → C → A, as shown in the figure. If the net heat supplied to the gas in the cycle is